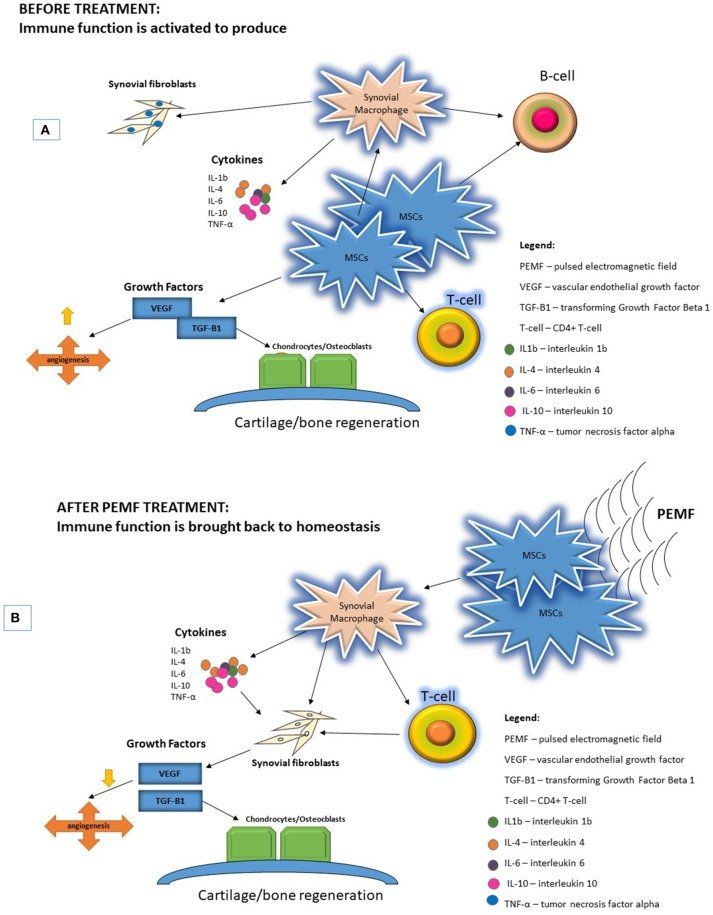
This is Part Four in a
series
on PEMF or Pulsed Electromagnetic Field Therapy, which is a modality we are pleased to offer in addition to
Frequency Specific Microcurrent right here at The Healing Collective.
WHAT IS PEMF?
Pulsed Electro-Magnetic Field Therapy is a noncontact, noninvasive, non-pharmacological modality that is used to promote optimal cellular function so that your body can do what it is designed to do: heal.
Click here for our introductory post on PEMF.
PEMF and associated therapies do not claim to diagnose, treat or cure any disease.
When we first started to dive into the published literature on Pulsed Electromagnetic Field Therapy (PEMF), we were surprised to learn of its long history and the sheer volume of data that has been published. From historic bone healing (which has been studied since the 1800s) to depression, disc pain to water treatment options, there is a trove of research to explore.
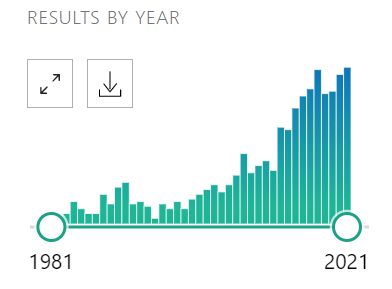
This post is an attempt to offer some brief, abstracted glimpses into just some of the data available on PEMF and is meant to be a jumping-off point for further and deeper study for those interested. Below, you will find a references list organized by date of publication that includes notable findings from the study abstracts.
One way you might choose to engage is to use the Search feature on your keyboard (Ctrl + F on a laptop or Command + F on a Mac) and see if there is an article that addresses a condition you are curious about. Or, simply start browsing the list below. This list is far from inclusive so please excuse any omissions; we will add to it as we are able.
THE DATA (BY YEAR) & SUMMARY
2021
Bademoğlu G, Erdal N, Uzun C, Taşdelen B.
The effects of pulsed electromagnetic field on experimentally induced sciatic nerve injury in rats. Electromagn Biol Med. 2021; 40(3):408-419. doi:10.1080/15368378.2021.1907403
ABSTRACT: “Some experimental research indicates that low-frequency pulsed electromagnetic field (PEMF) stimulation may accelerate regeneration in sciatic nerve injury. However, little research has examined the electrophysiological and functional properties of regenerating peripheral nerves under PEMF. The main aim of the present study is to investigate the effects of PEMF on sciatic nerve regeneration in short- and long-term processes with electrophysiologically and functionally after crushing damage. Crush lesions were performed using jewelery forceps for 30 s. After crush injury of the sciatic nerves, 24 female Wistar-Albino rats were divided into 3 groups with 8 rats in each group: SH(Sham), SNI (Sciatic Nerve Injury), SNI+PEMF(Sciatic Nerve Injury+Pulsed Electromagnetic Field). SNI+PEMF group was exposed to PEMF (4 h/day, intensity; 0.3mT, low-frequency; 2 Hz) for 40-days. Electrophysiological records (at the beginning and 1st, 2nd, 4th and 6th weeks post-crush) and functional footprints (at 1st, 2nd, 3rd, 4th, 5th and 6th weeks post crush) were measured from all groups during the experiment. The results were compared to SNI and SNI+PEMF groups, it was found that amplitude and area parameters in the first-week were significantly higher and latency was lower in the SNI+PEMF group than in the SNI group (p < 0,05). However, the effect of PEMF was not significant in the 2nd, 4th, 6th weeks. In addition, in the 1st and 2nd weeks, the SSI parameters were significantly higher in SNI+PMF group than SNI group (p < .05). These results indicate that low-frequency PEMF is not effective for long-periods of application time while PEMF may be useful during the short-term recovery period.”
Celik C, Franco-Obregón A, Lee EH, Hui JH, Yang Z.
Directionalities of magnetic fields and topographic scaffolds synergise to enhance MSC chondrogenesis. Acta Biomater. 2021;119:169-183. doi:10.1016/j.actbio.2020.10.039
ABSTRACT: “We have previously shown that brief, low-amplitude pulsed electromagnetic fields (PEMFs) differentially enhance mesenchymal stem cell (MSC) chondrogenesis in scaffold-free pellet cultures versus conventional tissue culture plastic (TCP), indicating an interplay between magnetism and micromechanical environment. Here, we examined the influence of PEMF directionality over the chondrogenic differentiation of MSCs laden on electrospun fibrous scaffolds of either random (RND) or aligned (ALN) orientations. Correlating MSCs' chondrogenic outcome to pFAK activation and YAP localisation, MSCs on the RND scaffolds experienced the least amount of resting mechanical stress and underwent greatest chondrogenic differentiation in response to brief PEMF exposure (10 min at 1 mT) perpendicular to the dominant plane of the scaffolds (Z-directed). By contrast, in MSC-impregnated RND scaffolds, greatest mitochondrial respiration resulted from X-directed PEMF exposure (parallel to the scaffold plane), and was associated with curtailed chondrogenesis. MSCs on TCP or the ALN scaffolds exhibited greater resting mechanical stress and accordingly, were unresponsive, or negatively responsive, to PEMF exposure from all directions. The efficacy of PEMF-induced MSC chondrogenesis is hence regulated in a multifaceted manner involving focal adhesion dynamics, as well as mitochondrial responses, culminating in a final cellular response. The combined contributions of micromechanical environment and magnetic field orientation hence will need to be considered when designing magnetic exposure paradigms.”
"PEMF plays
a role in enhancing angiogenesis, and its clinical application may go far beyond the current scope."
Peng L, Fu C, Wang L, Zhang, Q, Liang Z, He C and Wei Q.
The Effect of Pulsed Electromagnetic Fields on Angiogenesis. Bioelectromagnetics. 2021; 42: 250-258.
https://doi.org/10.1002/bem.22330
ABSTRACT: “A pulsed electromagnetic field (PEMF) has been used to treat inflammation-based diseases such as osteoporosis, neurological injury, and osteoarthritis. Numerous animal experiments and in vitro studies have shown that PEMF may affect angiogenesis. For ischemic diseases, in theory, blood flow may be richer by increasing the number of blood vessels which supply blood to ischemic tissue. PEMF plays a role in enhancing angiogenesis, and their clinical application may go far beyond the current scope. In this review, we analyzed and summarized the effects and possible mechanisms of PEMF on angiogenesis. Most studies have shown that PEMF with specific parameters can promote angiogenesis, which is manifested by an increased vascular growth rate and increased capillary density. The potential mechanisms consist of promoting vascular endothelial cell proliferation, migration, and tube formation, and increasing the expression level of vascular endothelial growth factor (VEGF), fibroblast growth factor 2 (FGF2), angiopoietin-2 (Ang-2), and other angiogenic growth factors. Additionally, PEMF has an impact on the activation of voltage-gated calcium channels (VGCC).”
Tabakan I, Yuvacı AU, Taştekin B, Öcal I, Pelit A.
The healing effect of pulsed magnetic field on burn wounds. Burns. 2021; S0305-4179(21)00154-6. doi:10.1016/j.burns.2021.06.001
ABSTRACT: “In this study, the effect of PEMF on cutaneous wound healing in an animal burn model was evaluated and the dose and duration of the magnetic field should be discussed for this effect to occur. Animals were divided into five groups including eight each (n = 40) (Groups 1, 2, 3, 4, 5).Group 1 was the control group; received no treatment after second-degree burn wound. Group 2 received daily wound care with saline. Group 3 received daily wound care with pomade containing mupirocin. Group 4 received Pulsed Electromagnetic Field signal for 60 min (1.5 m T and 40 Hz for seven days) and Group 5 also received PEMF signal for 60 min the same frequency and intensity for 14 days. Microscopically, second-degree burn wounds were successfully detected in all rats. Histopathological examination results in no significant difference between groups in neutrophil infiltration. The difference between the groups in vascularization was statistically significant between Group II and Group V (p < 0.001) and between Group I and Group V (p = 0.005) Epithelialization was present in 75% of the rats in Group V, while no epithelialization was observed in any of the other groups. In conclusion, we observed a significant improvement in the stasis zone of the group receiving Pulsed Electromagnetic Field for two weeks.”
Varani K, Vincenzi F, Pasquini S, et al.
Pulsed Electromagnetic Field Stimulation in Osteogenesis and Chondrogenesis: Signaling Pathways and Therapeutic Implications. Int J Mol Sci. 2021; 22(2):809. doi:10.3390/ijms22020809
ABSTRACT: “Mesenchymal stem cells (MSCs) are the main cell players in tissue repair and thanks to their self-renewal and multi-lineage differentiation capabilities, they gained significant attention as cell source for tissue engineering (TE) approaches aimed at restoring bone and cartilage defects. Despite significant progress, their therapeutic application remains debated: the TE construct often fails to completely restore the biomechanical properties of the native tissue, leading to poor clinical outcomes in the long term. Pulsed electromagnetic fields (PEMFs) are currently used as a safe and non-invasive treatment to enhance bone healing and to provide joint protection. PEMFs enhance both osteogenic and chondrogenic differentiation of MSCs. Here, we provide extensive review of the signaling pathways modulated by PEMFs during MSCs osteogenic and chondrogenic differentiation. Particular attention has been given to the PEMF-mediated activation of the adenosine signaling and their regulation of the inflammatory response as key player in TE approaches. Overall, the application of PEMFs in tissue repair is foreseen: (1) in vitro: to improve the functional and mechanical properties of the engineered construct; (2) in vivo: (i) to favor graft integration, (ii) to control the local inflammatory response, and (iii) to foster tissue repair from both implanted and resident MSCs cells.”
2020
Alzayed KA, Alsaadi SM.
Efficacy of Pulsed Low-Frequency Magnetic Field Therapy on Patients with Chronic Low Back Pain: A Randomized Double-Blind Placebo-Controlled Trial. Asian Spine J. 2020; 14(1):33-42. doi:10.31616/asj.2019.0043 RESULTS: “The treatment group experienced a more rapid improvement in both pain and disability compared with the placebo group. The analysis showed a significant improvement in the pain intensity and disability scores in the treatment group at week 3 (p < 0.05), whereas an improvement in the placebo group was detected at week 6. The significant improvement in both groups was sustained for weeks 6, 9, and 13. There was no difference between the groups in scores of pain intensity and disability at weeks 6 and 13.”
"Low-frequency magnetic therapy in complex rehabilitation contributes to the regression of respiratory disorders."
"PEMFs inhibit neuroinflammation in N9 cells ... and may be relevant to treat a variety of diseases characterized by neuroinflammation."
Merighi S, Gessi S, Bencivenni S, et al.
Signaling pathways involved in anti-inflammatory effects of Pulsed Electromagnetic Field in microglial cells. Cytokine. 2020; 125:154777. doi:10.1016/j.cyto.2019.154777
ABSTRACT: “PEMFs reduced the LPS-increased production of TNF-α and IL-1β in N9 cells, through a pathway involving JNK1/2. Furthermore, they decreased the LPS-induced release of IL-6, by a mechanism not dependent on AC, PLC, PKC-ε, PKC-δ, p38, ERK1/2, JNK1/2, Akt and caspase 1. Importantly, a significant effect of PEMFs in the reduction of crucial cell functions specific of microglia like ROS generation, cell invasion and phagocytosis was found. PEMFs inhibit neuroinflammation in N9 cells through a mechanism involving, at least in part, the activation of JNK MAPK signaling pathway and may be relevant to treat a variety of diseases characterized by neuroinflammation.”
Miller, CP, Prener, M, Dissing, S, Paulson, OB.
Transcranial low-frequency pulsating electromagnetic fields (T-PEMF) as post-concussion syndrome treatment. Acta Neurol Scand. 2020; 142: 597– 604.
https://doi.org/10.1111/ane.13300
RESULTS: “Compliance was high as all subject completed the full treatment. Two patients however experienced a worsening of their concussion symptoms during the course of treatment. The remaining patients had some discomfort in relation to treatment, mainly headache, but passing and less for each treatment. The majority (n = 5) had a reduction in symptoms overall, up to 61% (2%-61%) based on the Rivermead Post-Concussion Symptoms Questionnaire. Further studies on T-PEMF as a treatment option for post-concussion syndrome are warranted.”
Nayak BP, Dolkart O, Satwalekar P, et al. Effect of the Pulsed Electromagnetic Field (PEMF) on Dental Implants Stability: A Randomized Controlled Clinical Trial. Materials (Basel). 2020; 13(7):1667. Published 2020 Apr 3. doi:10.3390/ma13071667
ABSTRACT: “A pulsed electromagnetic field (PEMF) has been shown to contribute to heightening bone regeneration in a range of clinical areas, including dentistry. Due to the scarcity of studies using PEMF in oral implantology, the present experiment scrutinized the effect of PEMF can lead to improving the stability of the implant. A total of 19 subjects (40 implants in total) were selected to participate in the current study and were randomly allocated to either the PEMF group or control group. Subjects in the PEMF group received an activated miniaturized electromagnetic device (MED) while the control group received a sham healing cup. Implants stability was assessed by resonance frequency analyses (RFA) via implant stability quotient (ISQ) calculations. RFA were recorded as following: immediately after procedure, and then 2, 4, 6, 8 and 12 weeks later. Radiographic analysis was performed at baseline, 6 and 12 weeks after implant placement. Proinflammatory cytokines were evaluated in peri-implant crevicular fluid (PICF). The PEMF group presented higher ISQ mean values when compared to the control group. The primary stability time frame (the first 2 weeks) MED group depicted an increase in stability of 6.8%, compared to a decrease of 7.6% in the control group related to the baseline. An overall stability increase of 13% was found in MED treated group (p = 0.02), in contrast, the overall stability in the control group decreased by 2% (p = 0.008). TNF-α concentration during first 4 weeks was lower in the MED treated group. The data strongly suggests that MED generated continuing a PEMF may be considered as a new way to stimulate the stability of the implants at the early healing period.”
"Pulsed electromagnetic field has been shown to contribute to heightening bone regeneration in a range of clinical areas, including dentistry."
Parate D, Kadir ND, Celik C, et al.
Pulsed electromagnetic fields potentiate the paracrine function of mesenchymal stem cells for cartilage regeneration. Stem Cell Res Ther. 2020; 11(1):46. Published 2020 Feb 3. doi:10.1186/s13287-020-1566-5
ABSTRACT: “The findings reported here demonstrate that PEMF stimulation is capable of modulating the paracrine function of MSCs for the enhancement and re-establishment of cartilage regeneration in states of cellular stress. The PEMF-induced modulation of the MSC-derived paracrine function for directed biological responses in recipient cells or tissues has broad clinical and practical ramifications with high translational value across numerous clinical applications.”
Qiu XS, Li XG, Chen YX.
Pulsed electromagnetic field (PEMF): A potential adjuvant treatment for infected nonunion. Med Hypotheses. 2020;136:109506. doi:10.1016/j.mehy.2019.109506
ABSTRACT: “Infected nonunion is still a challenge for orthopaedic surgeons. The goal of treatment is to eliminate infection and achieve bone union. Surgery is the only effective method currently. However, it is invasive and the results are still unsatisfactory. Therefore, seeking a noninvasive and effective method to resolve infected nonunion is necessary. Pulsed electromagnetic field (PEMF) has been used for the treatment of nonunion for more than 40 years. PEMF could promote bone formation at tissue, cell and subcellular levels. Furthermore, our study showed that PEMF had bactericidal effect. The hypothesis we proposed herein is that PEMF may be an adjuvant treatment for infected nonunion by controlling infection and inducing bone formation.”
Ross C, Overholt T, Xu R, et al. Pulsed electromagnetic field (PEMF) as an adjunct therapy for pain management in interstitial cystitis/bladder pain syndrome [published online ahead of print, 2021 Jun 8]. Int Urogynecol J. 2021; 10.1007/s00192-021-04862-3. doi:10.1007/s00192-021-04862-3
ABSTRACT: “The use of PEMF has been evaluated as a therapeutic strategy for pain management in several clinical scenarios. Randomized, double-blinded, placebo-controlled trials have reported positive efficacy and safety profiles when PEMF was used to treat non-specific low back pain, patellofemoral pain syndrome, chronic post-operative pain, osteoarthritis-related pain, rheumatoid arthritis-related pain, and fibromyalgia-related pain. Based on these positive outcomes in a variety of pain conditions, clinical trials to evaluate whether PEMF can provide a safe, non-invasive therapeutic approach to improve symptoms of chronic pain and fatigue in patients with IC/BPS are warranted.”
Tai YK, Ng C, Purnamawati K et al.
Magnetic fields modulate metabolism and gut microbiome in correlation with Pgc-1α expression: Follow-up to an in vitro magnetic mitohormetic study. The FASEB Journal. 2020; 34: 11143– 11167.
https://doi.org/10.1096/fj.201903005RR
ABSTRACT: "The combination of PEMFs and exercise for 6 weeks enhanced running performance and upregulated muscular and adipose Pgc-1α transcript levels, whereas exercise alone was incapable of elevating Pgc-1α levels. The gut microbiome Firmicutes/Bacteroidetes ratio decreased with exercise and PEMF exposure, alone or in combination, which has been associated in published studies with an increase in lean body mass. After 2 months, brief PEMF treatment alone increased Pgc-1α and mitohormetic gene expression and after >4 months PEMF treatment alone enhanced oxidative muscle expression, fatty acid oxidation, and reduced insulin levels. Hence, short-term PEMF treatment was sufficient to instigate PGC-1α-associated transcriptional cascades governing systemic mitohormetic adaptations, whereas longer-term PEMF treatment was capable of inducing related metabolic adaptations independently of exercise."
Tenuta, M, Tarsitano, MG, Mazzotta, P, et al.
Therapeutic use of pulsed electromagnetic field therapy reduces prostate volume and lower urinary tract symptoms in benign prostatic hyperplasia. Andrology. 2020; 8: 1076–1085.
https://doi.org/10.1111/andr.12775
RESULTS: “PEMF was able to reduce PV after 28 days of therapy. Symptoms improved in a short time, with high compliance and no effects on hormonal and sexual function or any side effects. Patients with moderate-severe LUTS and without MetS seem to benefit more from this treatment.”
"PEMF therapy has clinically significant effects on pain in patients with osteoarthritis."
Yang X, He H, Ye W, Perry TA, He C.
Effects of Pulsed Electromagnetic Field Therapy on Pain, Stiffness, Physical Function, and Quality of Life in Patients With Osteoarthritis: A Systematic Review and Meta-Analysis of Randomized Placebo-Controlled Trials. Phys Ther. 2020;100(7):1118-1131. doi:10.1093/ptj/pzaa054
IMPACT: "Our study suggests that PEMF therapy has clinically significant effects on pain in patients with OA. The current evidence was limited to the short-term effects of PEMF therapy."
2019
Alvarez LX, McCue J, Lam NK, Askin G, Fox PR.
Effect of Targeted Pulsed Electromagnetic Field Therapy on Canine Postoperative Hemilaminectomy: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial. J Am Anim Hosp Assoc. 2019; 55(2):83-91. doi:10.5326/JAAHA-MS-6798
ABSTRACT: “The present prospective, double-blind, placebo-controlled trial evaluated targeted PEMF therapy administered to 53 client-owned dogs who underwent hemilaminectomy for naturally occurring disk extrusion intervertebral disc disease … Dogs receiving PEMF therapy following postoperative hemilaminectomy demonstrated improved wound scores at 6 wk and reduced mean number of owner-administered pain medications compared with the control group therapy.”
Cottrill E, Pennington Z, Ahmed AK, et al.
The effect of electrical stimulation therapies on spinal fusion: a cross-disciplinary systematic review and meta-analysis of the preclinical and clinical data. J Neurosurg Spine. 2019; 1-21. doi:10.3171/2019.5.SPINE19465
CONCLUSIONS: “The authors found that electrical stimulation devices may produce clinically significant increases in arthrodesis rates among patients undergoing spinal fusion. They also found that the pro-arthrodesis effects seen in preclinical studies are also found in clinical populations, suggesting that findings in animal studies are translatable. Additional research is needed to analyze the cost-effectiveness of these devices.”
Galli C, Pedrazzi G and Guizzardi S.
The cellular effects of Pulsed Electromagnetic Fields on osteoblasts: A review.
Bioelectromagnetics. 2019; 40: 211-233.
https://doi.org/10.1002/bem.22187
ABSTRACT: "Electromagnetic fields (EMFs) have long been known to interact with living organisms and their cells and to bear the potential for therapeutic use. Among the most extensively investigated applications, the use of Pulsed EMFs (PEMFs) has proven effective to ameliorate bone healing in several studies, although the evidence is still inconclusive. This is due in part to our still-poor understanding of the mechanisms by which PEMFs act on cells and affect their functions and to an ongoing lack of consensus on the most effective parameters for specific clinical applications. The present review has compared in vitro studies on PEMFs on different osteoblast models, which elucidate potential mechanisms of action for PEMFs, up to the most recent insights into the role of primary cilia, and highlight the critical issues underlying at least some of the inconsistent results in the available literature."
"A significant improvement was found in the neck pain, disability, depression, anxiety, and quality of life scores of both [physical therapy and PEMF] groups after treatment when compared to those before treatment."
ABSTRACT: "The finding that alterations in electrical potential play an important role in the mechanical stimulation of the bone provoked hype that noninvasive extremely low frequency pulsed electromagnetic fields (ELF-PEMF) can be used to support healing of bone and osteochondral defects. This resulted in the development of many ELF-PEMF devices for clinical use. Due to the resulting diversity of the ELF-PEMF characteristics regarding treatment regimen, and reported results, exposure to ELF-PEMFs is generally not among the guidelines to treat bone and osteochondral defects. Notwithstanding, here we show that there is strong evidence for ELF-PEMF treatment. We give a short, confined overview of in vitro studies investigating effects of ELF-PEMF treatment on bone cells, highlighting likely mechanisms. Subsequently, we summarize prospective and blinded studies, investigating the effect of ELF-PEMF treatment on acute bone fractures and bone fracture non-unions, osteotomies, spinal fusion, osteoporosis, and osteoarthritis. Although these studies favor the use of ELF-PEMF treatment, they likewise demonstrate the need for more defined and better controlled/monitored treatment modalities. However, to establish indication-oriented treatment regimen, profound knowledge of the underlying mechanisms in the sense of cellular pathways/events triggered is required, highlighting the need for more systematic studies to unravel optimal treatment conditions."
"Our findings suggest a potential role for the pulsed EMF for the amelioration of cognition impairment."
Li Y, Zhang Y, Wang W, et al.
Effects of pulsed electromagnetic fields on learning and memory abilities of STZ-induced dementia rats. Electromagn Biol Med. 2019; 38(2):123-130. doi:10.1080/15368378.2019.1591437
ABSTRACT: “Our findings indicate that the pulsed EMF exposure can improve the ability of learning and memory in STZ-induced dementia rats and this effect may be related to the process of IGF signal transduction, suggesting a potential role for the pulsed EMF for the amelioration of cognition impairment.”
Malling ASB, Morberg BM, Wermuth L, Gredal O, Bech P, Jensen BR.
The effect of 8 weeks of treatment with transcranial pulsed electromagnetic fields on hand tremor and inter-hand coherence in persons with Parkinson's disease. J Neuroeng Rehabil. 2019; 16(1):19. Published 2019 Jan 31. doi:10.1186/s12984-019-0491-2
ABSTRACT: “The T-PEMF treatment decreased the inter-hand coherence in the PD group with unilateral postural tremor. The PD group with unilateral postural tremor was less clinically affected by the disease than the PD group with bilateral postural tremor. However, no differences between T-PEMF and placebo treatment on either intensity related or coherence related measures were found when all persons with PD were included in the analyses. The peak power decreased and the tremor intensity tended to decrease in both treatment groups.”
Miyamoto H, Sawaji Y, Iwaki T, Masaoka T, Fukada E, Date M and Yamamoto K.
Intermittent pulsed electromagnetic field stimulation activates the mTOR pathway and stimulates the proliferation of osteoblast-like cells. Bioelectromagnetics. 2019; 40: 412-421.
https://doi.org/10.1002/bem.22207
ABSTRACT: “Continuous PEMF stimulation induced a transient phosphorylation of the mTOR pathway, whereas intermittent PEMF stimulation (1 cycle of 10 min stimulation followed by 20 min of stimulation pause) revitalized the reduced phosphorylation. Moreover, PEMF stimulation stimulated cell proliferation (bromodeoxyuridine incorporation) rather than differentiation (alkaline phosphatase activity), with a more notable effect in the intermittently stimulated cells. These results suggest that intermittent PEMF stimulation may be effective in promoting bone fracture healing by accelerating cell proliferation, and in shortening stimulation time.”
Mohammad Alizadeh MA, Abrari K, Lashkar Blouki T, Ghorbanian MT, Jadidi M.
Pulsed electromagnetic field attenuated PTSD-induced failure of conditioned fear extinction. Iran J Basic Med Sci. 2019; 22(6):650-659. doi:10.22038/ijbms.2019.32576.7797
RESULTS: “Using 14-day PEM attenuates the PTSD-induced failure of conditioned fear extinction and exaggerated sensitized fear, and this might be related to the neuroprotective effects of magnetic fields on the hippocampus.”
Ross CL, Ang DC, Almeida-Porada G. Targeting Mesenchymal Stromal Cells/Pericytes (MSCs) With Pulsed Electromagnetic Field (PEMF) Has the Potential to Treat Rheumatoid Arthritis. Front Immunol. 2019; 10:266. doi:10.3389/fimmu.2019.00266
ABSTRACT: “Rheumatoid arthritis (RA) is a systemic autoimmune disease characterized by chronic inflammation of synovium (synovitis), with inflammatory/immune cells and resident fibroblast-like synoviocytes (FLS) acting as major players in the pathogenesis of this disease. The resulting inflammatory response poses considerable risks as loss of bone and cartilage progresses, destroying the joint surface, causing joint damage, joint failure, articular dysfunction, and pre-mature death if left untreated. At the cellular level, early changes in RA synovium include inflammatory cell infiltration, synovial hyperplasia, and stimulation of angiogenesis to the site of injury. Different angiogenic factors promote this disease, making the role of anti-angiogenic therapy a focus of RA treatment. To control angiogenesis, mesenchymal stromal cells/pericytes (MSCs) in synovial tissue play a vital role in tissue repair.
"These results further establish PEMFs anti-inflammatory activity, and may inform potential future clinical uses for management of inflammation associated with disc degeneration."
Tang X, Coughlin D, Ballatori A, et al.
Pulsed Electromagnetic Fields Reduce Interleukin-6 Expression in Intervertebral Disc Cells Via Nuclear Factor-κβ and Mitogen-Activated Protein Kinase p38 Pathways. Spine (Phila Pa 1976). 2019; 44(22):E1290-E1297. doi:10.1097/BRS.0000000000003136
CONCLUSION: “Our data indicate that the previously-reported inhibitory effect of PEMF treatment on disc inflammation is mediated by NF-κβ and phosphorylated p38-MAPK signaling pathways. These results further establish PEMFs anti-inflammatory activity, and may inform potential future clinical uses for management of inflammation associated with disc degeneration.”
2018
Gaynor JS, Hagberg S, Gurfein BT.
Veterinary applications of pulsed electromagnetic field therapy. Res Vet Sci. 2018; 119:1-8. doi:10.1016/j.rvsc.2018.05.005
ABSTRACT: “Pulsed electromagnetic field (PEMF) therapy can non-invasively treat a variety of pathologies by delivering electric and magnetic fields to tissues via inductive coils. The electromagnetic fields generated by these devices have been found to affect a variety of biological processes and basic science understanding of the underlying mechanisms of action of PEMF treatment has accelerated in the last 10 years. Accumulating clinical evidence supports the use of PEMF therapy in both animals and humans for specific clinical indications including bone healing, wound healing, osteoarthritis and inflammation, and treatment of post-operative pain and edema. While there is some confusion about PEMF as a clinical treatment modality, it is increasingly being prescribed by veterinarians. In an effort to unravel the confusion surrounding PEMF devices, this article reviews important PEMF history, device taxonomy, mechanisms of action, basic science and clinical evidence, and relevant trends in veterinary medicine. The data reviewed underscore the usefulness of PEMF treatment as a safe, non-invasive treatment modality that has the potential to become an important stand-alone or adjunctive treatment modality in veterinary care.”
Sorrell RG, Muhlenfeld J, Moffett J, Stevens G, Kesten S. Evaluation of pulsed electromagnetic field therapy for the treatment of chronic postoperative pain following lumbar surgery: a pilot, double-blind, randomized, sham-controlled clinical trial. J Pain Res. 2018; 11:1209-1222. Published 2018 Jun 22. doi:10.2147/JPR.S164303
RESULTS: “PEMF therapy (42-μs pulse width) was associated with trends for a reduction in pain, compared to sham treatment. Secondary endpoints were consistent with an overall beneficial effect of the PEMF 42-μs pulse width device.”
Huegel J, Choi DS, Nuss CA, et al.
Effects of pulsed electromagnetic field therapy at different frequencies and durations on rotator cuff tendon-to-bone healing in a rat model. J Shoulder Elbow Surg. 2018; 27(3):553-560. doi:10.1016/j.jse.2017.09.024
RESULTS: "Overall, our results suggest that PEMF therapy has a positive effect on rat rotator cuff healing for each electromagnetic fundamental pulse frequency and treatment duration tested in this study."
"Our data suggest that PEMF may have a role in the clinical management of patients with chronic low back pain."
Tang X, Alliston T, Coughlin D, Miller S, Zhang N, Waldorff EI, Ryaby JT and Lotz JC.
Dynamic imaging demonstrates that pulsed electromagnetic fields (PEMF) suppress IL-6 transcription in bovine nucleus pulposus cells. J Orthop Res. 2018; 36: 778-787.
https://doi.org/10.1002/jor.23713
ABSTRACT: “Imaging and PCR data demonstrated that the inductive effect of IL-1α on IL-6 expression could be significantly inhibited by PEMF treatment in a time-dependent manner (early as 2 h of stimulus initiation). Our data suggest that PEMF may have a role in the clinical management of patients with chronic low back pain. Furthermore, this study shows that the MS2-tagged GFP reporter system is a useful tool for visualizing the dynamic events of mechanobiology in musculoskeletal research.”
We're just getting started with some of the most recent data ...
Many many more studies to come ... we will update this page frequently, so check back soon.
Currently, Dr. Jessie integrates PEMF therapies into
Physical Therapy and Frequency Specific Microcurrent sessions that take into account your unique history and clinical picture. You can contact us to schedule a free 15 minute consult to learn more.
Subscribe to our Blog and get updates on
PEMF Therapy straight to your inbox: